

Stevanato Group Q4 and FY 2025 Financial Results March 4, 2026 Exhibit 99.1

Q4 and FY 2025 Financial Results Safe Harbor Statement Forward-Looking Statements This presentation contains certain forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 that reflect the current views of Stevanato Group S.p.A. (“we”, “our”, “us”, “Stevanato Group” or the “Company”) and which involve known and unknown risks, uncertainties and assumptions because they relate to events and depend on circumstances that will occur in the future whether or not within the control of the Company. These forward-looking statements include, or may include words such as "continued," “remain,” "growing," "rising," "expect," “growth," "continue," "position," "assumes," "will be," "expected," "expanding," "drive," and other similar terminology. Forward-looking statements contained in this presentation include, but are not limited to, statements about: our future financial performance, including our revenue, operating expenses and our ability to maintain profitability and operational and commercial capabilities; our expectations regarding the development of our industry and the competitive environment in which we operate; the expansion of our plants and sites, and our expectations related to our capacity expansion; the global supply chain and our committed orders; customer demand; the success of the Company's initiatives to optimize the industrial footprint, harmonize processes and enhance supply chain and logistics strategies; our geographical and industrial footprint; and our goals, strategies, and investment plans. These statements are neither promises nor guarantees but involve known and unknown risks, uncertainties and other important factors and circumstances that may cause Stevanato Group’s actual results, performance or achievements to be materially different from its expectations expressed or implied by the forward-looking statements, including conditions of the U.S. capital markets, negative global and domestic economic and political conditions, inflation, the impact of the conflict between Russia and Ukraine, the evolving events in Israel and Gaza, supply chain and logistical challenges and other negative developments affecting Stevanato Group’s business or unfavorable legislative or regulatory developments. The following are some of the factors that could cause our actual results to differ materially from those expressed in or underlying our forward-looking statements: (i) our product offerings are highly complex, and, if our products do not satisfy applicable quality criteria, specifications and performance standards, we could experience lost sales, delayed or reduced market acceptance of our products, increased costs and damage to our reputation; (ii) we must develop new products and enhance existing products, adapt to significant technological and innovative changes and respond to introductions of new products by competitors to remain competitive; (iii) if we fail to maintain and enhance our brand and reputation, our business, results of operations and prospects may be materially and adversely affected; (iv) we are highly dependent on our management and employees. Competition for our employees is intense, and we may not be able to attract and retain the highly skilled employees that we need to support our business and our intended future growth; (v) our business, financial condition and results of operations depend upon maintaining our relationships with suppliers and service providers; (vi) our business, financial condition and results of operations depend upon the availability and price of high-quality materials and energy supply and our ability to contain production costs; (vii) significant interruptions in our operations could harm our business, financial condition and results of operations; (viii) as a consequence of the COVID-19 pandemic, sales of vials to and for vaccination programs globally increased resulting in a revenue growth acceleration. The demand for such products may shrink, as the need for COVID-19 related solutions continue to decline; (ix) our manufacturing facilities are subject to operating hazards which may lead to production curtailments or shutdowns and have an adverse effect on our business, results of operations, financial condition or cash flows; (x) our business, financial condition and results of operations may be impacted by our ability to successfully expand capacity to meet customer demand ; (xi) the loss of a significant number of customers or a reduction in orders from a significant number of customers, including through destocking initiatives or lack of transparency of our products held by customers, could reduce our sales and harm our financial performance; (xii) we may face significant competition in implementing our strategies for revenue growth in light of actions taken by our competitors; (xiii) our global operations are subject to international market risks that may have a material effect on our liquidity, financial condition, results of operations and cash flows; (xiv) we are required to comply with a wide variety of laws and regulations and are subject to regulation by various federal, state and foreign agencies; (xv) given the relevance of our activities in the healthcare sector, investments by non-Italian entities in the Company, as well as certain asset disposals by the Company, may be subject to the prior authorization of the Italian Government (so called “golden powers”); (xvi) if relations between China and the U.S. deteriorate (including in connection with the current trade policy of the U.S. government), our business in the U.S. and China could be materially and adversely affected; (xvii) the U.S. government recently has implemented tariffs on certain products manufactured in several jurisdictions, including among others China, Mexico, the European Union and other European countries, and Canada, and has made announcements regarding the potential modification of existing tariffs and imposition of tariffs on other jurisdictions. Such tariffs as well as other trade policies that the U.S. government may implement in the future and the restrictive trade measures that other countries may adopt in response thereto, could adversely affect our business by making it more difficult or costly to trade goods between different jurisdictions; (xviii) cyber security risks and the failure to maintain the confidentiality, integrity and availability of our computer hardware, software and internet applications and related tools and functions, could result in damage to our reputation, data integrity and/or subject us to costs, fines or lawsuits under data privacy or other laws or contractual requirements; (xix) our trade secrets may be misappropriated or disclosed, and confidentiality agreements with directors, employees and third parties may not adequately prevent disclosure of trade secrets and protect other proprietary information; (xx) if we are unable to obtain and maintain patent protection for our technology, products and potential products, or if the scope of the patent protection obtained is not sufficiently broad, we may not be able to compete effectively in our markets; (xxi) we depend in part on proprietary technology licensed from others, and if we lose our existing licenses or are unable to acquire or license additional proprietary rights from third parties, we may not be able to continue developing our potential products; and (xxii) we are obligated to maintain proper and effective internal control over financial reporting. Our internal controls were not effective for the year ended December 31, 2025, and in the future may not be determined to be effective, which may adversely affect investor confidence in us and, as a result, the value of our ordinary shares; and any other risk described under the headings “Risk Factors,” “Operating and Financial Review and Prospects” and “Business” in our most recent Annual Report on Form 20-F filed with the U.S. Securities and Exchange Commission. This list is not exhaustive. We therefore caution you against relying on these forward-looking statements and we qualify all of our forward-looking statements by these cautionary statements. These forward-looking statements speak only as at their dates. The Company undertakes no obligation to update any forward-looking statement or statements to reflect events or circumstances after the date on which such statement is made or to reflect the occurrence of unanticipated events. New factors emerge from time to time, and it is not possible to predict all of these factors. Further, the Company cannot assess the impact of each such factor on our business or the extent to which any factor, or combination of factors, may cause actual results to be materially different from those contained in any forward-looking statements. For a description of certain additional factors that could cause the Company’s future results to differ from those expressed in any such forward-looking statements, refer to the risk factors discussed in our most recent Annual Report on Form 20-F filed with the U.S. Securities and Exchange Commission. Non-GAAP Financial Information This presentation contains non-GAAP financial measures. Please refer to the tables included in this presentation for a reconciliation of non-GAAP financial measures. Management monitors and evaluates its operating and financial performance using several non-GAAP financial measures, including Constant Currency Revenue, EBITDA, Adjusted EBITDA, Adjusted EBITDA Margin, Adjusted Operating Profit, Adjusted Operating Profit Margin, Adjusted Income Taxes, Adjusted Net Profit, Adjusted Diluted EPS, Capital Employed, Net Cash, Free Cash Flow and CAPEX. The Company believes that these non-GAAP financial measures provide useful and relevant information regarding its performance and improve its ability to assess its financial condition. While similar measures are widely used in the industry in which the Company operates, the financial measures it uses may not be comparable to other similarly titled measures used by other companies, nor are they intended to be substitutes for measures of financial performance or financial position as prepared in accordance with IFRS. Accordingly, you should not place undue reliance on any non-IFRS financial measures contained in this presentation.

Q4 and FY 2025 Financial Results Stevanato Group Q4 and FY 2025 Financial Results Earnings Call Franco Stevanato Chairman & CEO Marco Dal Lago CFO Lisa Miles CCO and IR

Franco Stevanato Chairman & Chief Executive Officer Q4 and FY 2025 Financial Results

Q4 and FY 2025 Financial Results FY25: Top-line Growth, Record Mix of HVS, and Continued Margin Expansion Total Company revenue increased 9% at constant currency (7% on a reported basis) for FY 2025 Biopharmaceutical and Diagnostic Solutions Segment (BDS) delivered DD top-line growth, offsetting the expected revenue decline from Engineering Segment HVS primary driver of revenue growth and margin expansion, executing our strategy set at the time of IPO Continuing to move up the value chain, and pivoting away from certain non-high-value categories not aligned with our strategy Continuing to execute our strategic priorities, including aligning growth investments in our business with customer demand trends HVS grew +29% yoy in 2025, on a full year basis Main driver for margin expansion Committed to meeting customer demand for HVS, expanding capacity in in Fishers and Latina Continued Strong Growth from HVS 2019 2024 2025 (Revenues from HVS as a % of Total Company Revenues) Ended 2025 With Another Solid Quarter Leading To Positive Full‑Year Performance And Strong Momentum As We Start 2026 __________ All comparisons refer to 2024 unless otherwise specified.
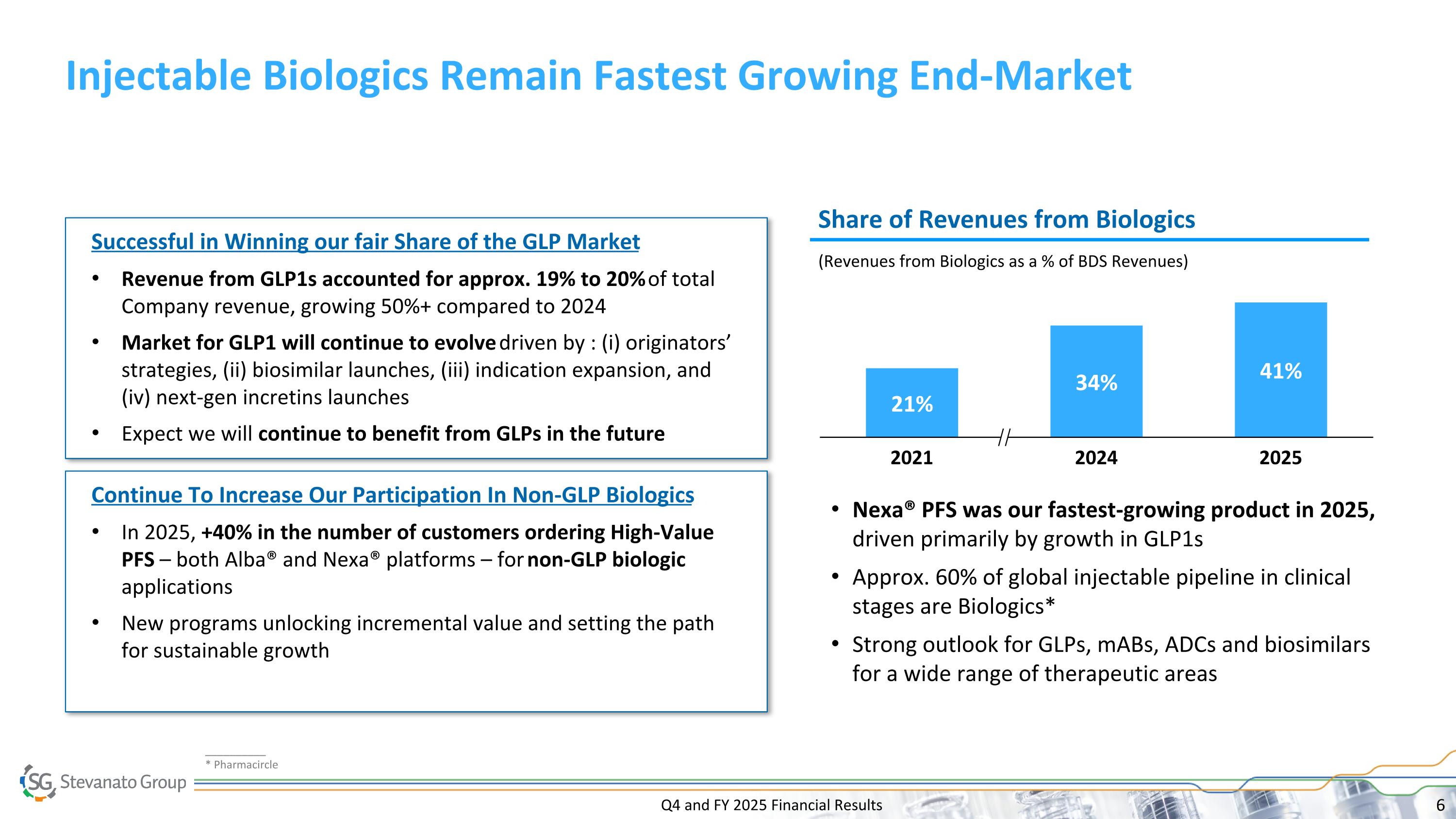
Q4 and FY 2025 Financial Results Injectable Biologics Remain Fastest Growing End-Market 2021 2024 2025 Successful in Winning our fair Share of the GLP Market Revenue from GLP1s accounted for approx. 19% to 20% of total Company revenue, growing 50%+ compared to 2024 Market for GLP1 will continue to evolve driven by : (i) originators’ strategies, (ii) biosimilar launches, (iii) indication expansion, and (iv) next-gen incretins launches Expect we will continue to benefit from GLPs in the future Share of Revenues from Biologics (Revenues from Biologics as a % of BDS Revenues) Continue To Increase Our Participation In Non-GLP Biologics In 2025, +40% in the number of customers ordering High-Value PFS – both Alba® and Nexa® platforms – for non-GLP biologic applications New programs unlocking incremental value and setting the path for sustainable growth Nexa® PFS was our fastest-growing product in 2025, driven primarily by growth in GLP1s Approx. 60% of global injectable pipeline in clinical stages are Biologics* Strong outlook for GLPs, mABs, ADCs and biosimilars for a wide range of therapeutic areas __________ * Pharmacircle

Q4 and FY 2025 Financial Results Latina, Italy Scaling commercial production for high-value Nexa® PFS Installation of PFS manufacturing lines and validation activities will continue in 2026, as planned The next phase devoted to increasing capacity for RTU EZ-fill® cartridges to meet the rising global demand Fishers (IN), U.S. In 2025 focused on Core Activities - line installations and validation Doubled the number of customers that are now validated Several PFS lines running commercial production Advancing device contract manufacturing buildout; still expect commercial activities to begin at the end of 2026 or early 2027 Supporting U.S. customers across the full value chain, strategically focused to meet demand for biologics Diversifying EMEA footprint with expanding capacity for PFS and EZ-fill® cartridges to satisfy market demand Demand-driven Capacity Expansion Projects to Support Growth in Biologics

Q4 and FY 2025 Financial Results Engineering Segment: Progress Update on Optimization Plan Focused 2025 efforts on advancing the operations Optimization Plan (i) right-sizing operations, (ii) streamlining processes, (iii) increasing standardization, and (iv) strengthening internal functions (eg, Project Management Office) Consolidated offices in Denmark, moved Visual Inspection activities to Italy, acquired a new site in Bologna – automation technology hub Action contributed to DD yoy growth in customer Site Acceptance Tests (SATs) 2026 guide assumes revenue decrease due to low order intake in prior months and slow pace of opportunity conversion into firm orders persists Sharply focused our sales & marketing efforts, which has led to a more robust opportunity pipeline, particularly in Visual Inspection Efforts Designed To Drive Efficiency And Productivity Gains To Best Position The Segment For Long-term Success.

Marco Dal Lago Chief Financial Officer Q4 and FY 2025 Financial Results

Q4 and FY 2025 Financial Results Q4 2025: Financial Highlights __________ All comparisons refer to Q4 2024 unless otherwise specified. * Adjusted operating profit margin, adjusted net profit, adjusted DEPS, adjusted EBITDA, adjusted EBITDA margin, are non-GAAP financial measures. Please refer to slides 17 to 22 for a reconciliation of non-GAAP measures (€ Million) Q4 2025: Revenue Q4 2025: Margins 200 131 (40%) Q4 2024 175 171 (49%) Q4 2025 331 346 +31% +5% HVS non-HVS Gross profit margin increased 120 bps to 30.9%, driven by: Favorable mix of accretive HVS Improvements from Latina and Fishers as we scale production (sites remain margin-dilutive) Continued recovery in vial demand Positive trends were partially offset by tariffs and unfavorable impact of FX Operating profit margin was 20.2% in line with previous year (adj. operating profit margin was 21.1%) Net profit of €47.6M, or €0.17 of diluted EPS (adj. net profit* of €49.8M, or €0.18 of adjusted diluted EPS*) Adjusted EBITDA* increased 7% to €97.7M; adjusted EBITDA margin* increased 70 bps to 28.2% Revenue increase driven by 10% growth in the BDS Segment (13% at CC), which offset the expected decline in the Engineering Segment HVS increased 31% yoy to €171.4 M and represented 49% of total revenue driven by strong demand in high-value PFS, and to a lesser extent, EZ-fill® cartridges +7% +35% growth at CC
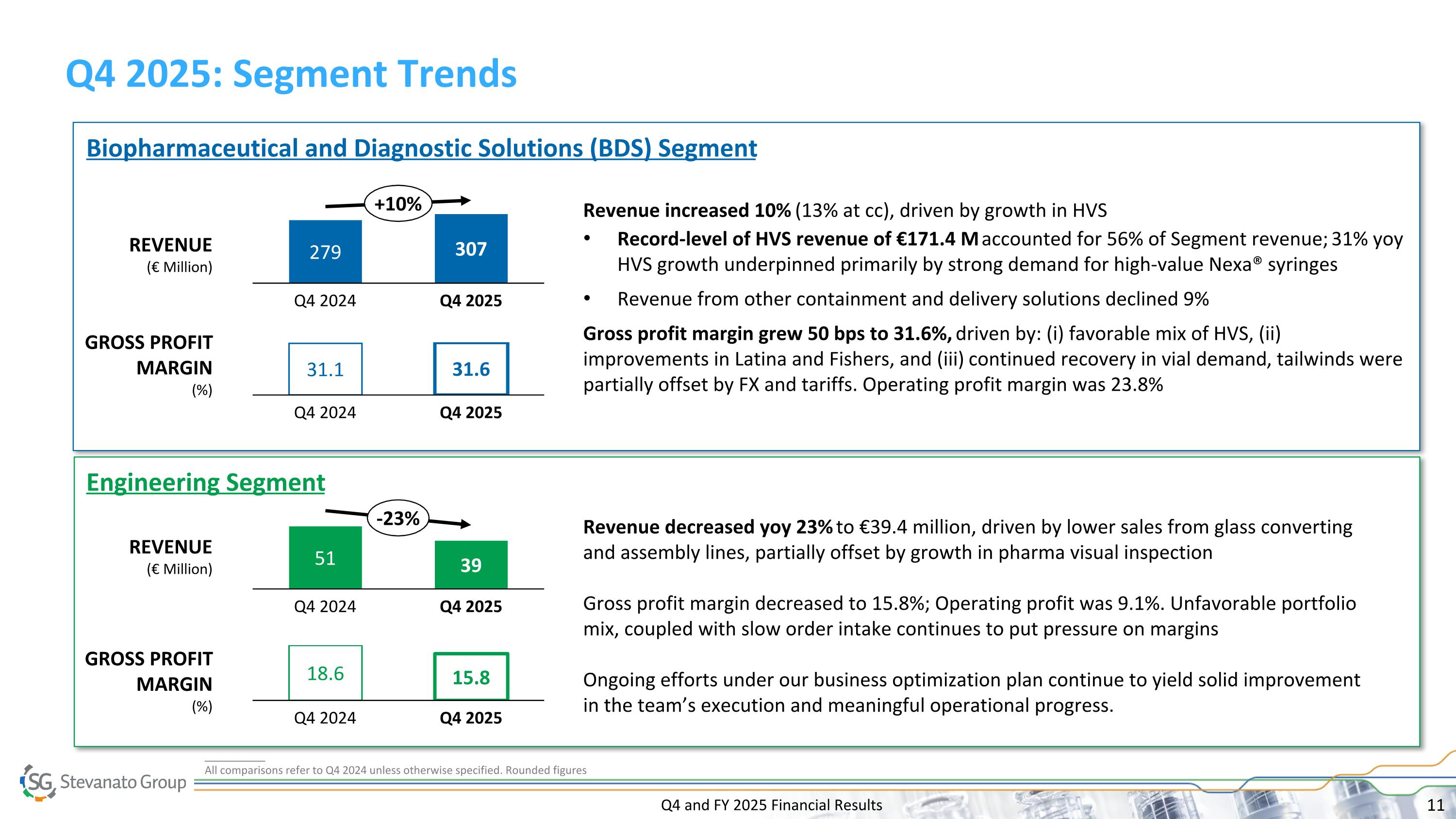
Q4 and FY 2025 Financial Results Q4 2025: Segment Trends 279 Q4 2024 307 Q4 2025 +10% Revenue increased 10% (13% at cc), driven by growth in HVS Record-level of HVS revenue of €171.4 M accounted for 56% of Segment revenue; 31% yoy HVS growth underpinned primarily by strong demand for high-value Nexa® syringes Revenue from other containment and delivery solutions declined 9% Gross profit margin grew 50 bps to 31.6%, driven by: (i) favorable mix of HVS, (ii) improvements in Latina and Fishers, and (iii) continued recovery in vial demand, tailwinds were partially offset by FX and tariffs. Operating profit margin was 23.8% Biopharmaceutical and Diagnostic Solutions (BDS) Segment REVENUE (€ Million) GROSS PROFIT MARGIN (%) Engineering Segment 31.1 Q4 2024 31.6 Q4 2025 51 Q4 2024 39 Q4 2025 -23% Revenue decreased yoy 23% to €39.4 million, driven by lower sales from glass converting and assembly lines, partially offset by growth in pharma visual inspection Gross profit margin decreased to 15.8%; Operating profit was 9.1%. Unfavorable portfolio mix, coupled with slow order intake continues to put pressure on margins Ongoing efforts under our business optimization plan continue to yield solid improvement in the team’s execution and meaningful operational progress. REVENUE (€ Million) GROSS PROFIT MARGIN (%) 18.6 Q4 2024 15.8 Q4 2025 __________ All comparisons refer to Q4 2024 unless otherwise specified. Rounded figures
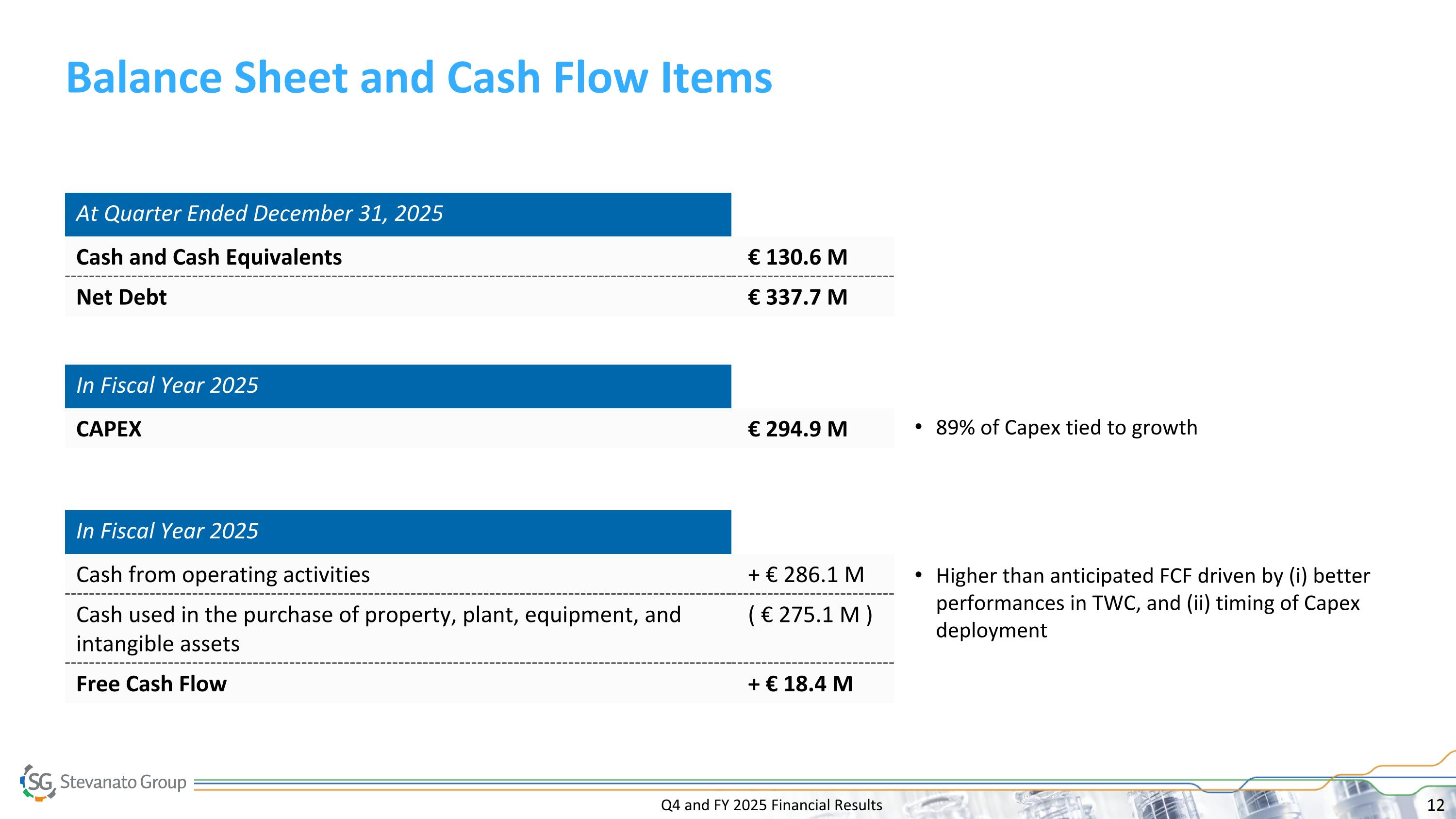
Q4 and FY 2025 Financial Results Balance Sheet and Cash Flow Items In Fiscal Year 2025 Cash from operating activities + € 286.1 M Cash used in the purchase of property, plant, equipment, and intangible assets ( € 275.1 M ) Free Cash Flow + € 18.4 M In Fiscal Year 2025 CAPEX € 294.9 M At Quarter Ended December 31, 2025 Cash and Cash Equivalents € 130.6 M Net Debt € 337.7 M 89% of Capex tied to growth Higher than anticipated FCF driven by (i) better performances in TWC, and (ii) timing of Capex deployment

Q4 and FY 2025 Financial Results FY 2026 Guidance __________ *Adjusted operating profit margin, adjusted net profit, adjusted DEPS, adjusted EBITDA and adjusted EBITDA margin, Net Debt, CapEx, Free Cash Flow are non-GAAP financial measures. Please refer to slides 17 to 22 for a reconciliation of non-GAAP measures. FY 2026 Guidance Implied Growth Revenue € 1.260B - € 1.290B 6% to 9% Revenue at Constant Currency € 1.278B - € 1.308B 8% to 10% Adjusted DEPS* € 0.59 - € 0.63 8% to 16% Adjusted EBITDA* € 331.8M - € 346.9M 11% to 16% FY 2026 Guidance assumes: The second half of 2026 will be stronger than the first half FX headwind of approx. €18 million, with the greatest impact in Q1 (approx. €10 million). As a result, in 1Q26 we expect MSD revenue growth on a reported basis (based on mid-point of guide). BDS Segment is expected to grow HSD to LDD, while Engineering Segment is expected to decline MSD to LDD HVS in the range of approximately 47% to 48% of total 2026 revenue Weighted shares outstanding of approximately 273.1 million Total capital expenditures in the range of €270M to €290M; net of customer contribution, capex between €240M to €260M Free cash flow neutral to €20M positive on full year basis Tax rate of approx. 26.8%

Franco Stevanato Chairman & Chief Executive Officer Q4 and FY 2025 Financial Results

Q4 and FY 2025 Financial Results Remain Focused On Executing Our Key Priorities, Supported By Strong Business Fundamentals We operate in attractive, growing end markets with favorable secular tailwinds Strong innovation advancing patient care (i) delivering novel biologics supporting new therapeutic areas, such as obesity, (ii) expanding global access, and (iii) improving standards of care 9.000+ injectable drug products in the global pipeline in clinical and registration phases, and more than 60% are biologics We believe we are well positioned to serve this demand through our integrated value proposition, differentiated portfolio, and commitment to science‑driven innovation Biologics expected to remain a key driver of topline growth and margin expansion, along with operational improvements, as we continue to benefit from new capacity, productivity gains, and improvements within our Engineering segment Together, these efforts position us well to deliver long‑term, sustainable growth and shareholder value

caterina.tripepi@stevanatogroup.com taylor.gerrells@teamlewis.com (U.S. Media) lisa.miles@stevanatogroup.com giacomo.guiducci@stevanatogroup.com Investor Relations Contacts Media Relations Contacts Upcoming Conferences and Marketing Activities Bank of America Injectable Drug Package call series – March 9, 2026 (virtual) KeyBanc Capital Markets Healthcare Forum – March 17, 2026 (virtual)

This presentation contains non-GAAP financial measures. Please refer to the tables included in this presentation for a reconciliation of non-GAAP measures. Management monitors and evaluates our operating and financial performance using several non-GAAP financial measures, including Constant Currency Revenue, EBITDA, Adjusted EBITDA, Adjusted EBITDA Margin, Adjusted Operating Profit, Adjusted Operating Profit Margin, Adjusted Income Taxes, Adjusted Net Profit, Adjusted Diluted EPS, Capital Employed, Net Cash, Free Cash Flow, and CapEx. We believe that these non-GAAP financial measures provide useful and relevant information regarding our performance and improve our ability to assess our financial condition. While similar measures are widely used in the industry in which we operate, the financial measures we use may not be comparable to other similarly titled measures used by other companies, nor are they intended to be substitutes for measures of financial performance or financial position as prepared in accordance with IFRS. Reconciliation of Non-GAAP Financial Measures Q4 and FY 2025 Financial Results

Q4 and FY 2025 Financial Results Reconciliation of Non-GAAP Financial Measures (1/5) Reconciliation of EBITDA (Amounts in € millions) Calculation of Net Profit margin, Operating Profit Margin, Adjusted EBITDA Margin and Adjusted Operating Profit Margin (Amounts in € millions) Reconciliation of Revenue to Constant Currency Revenue (Amounts in € millions)
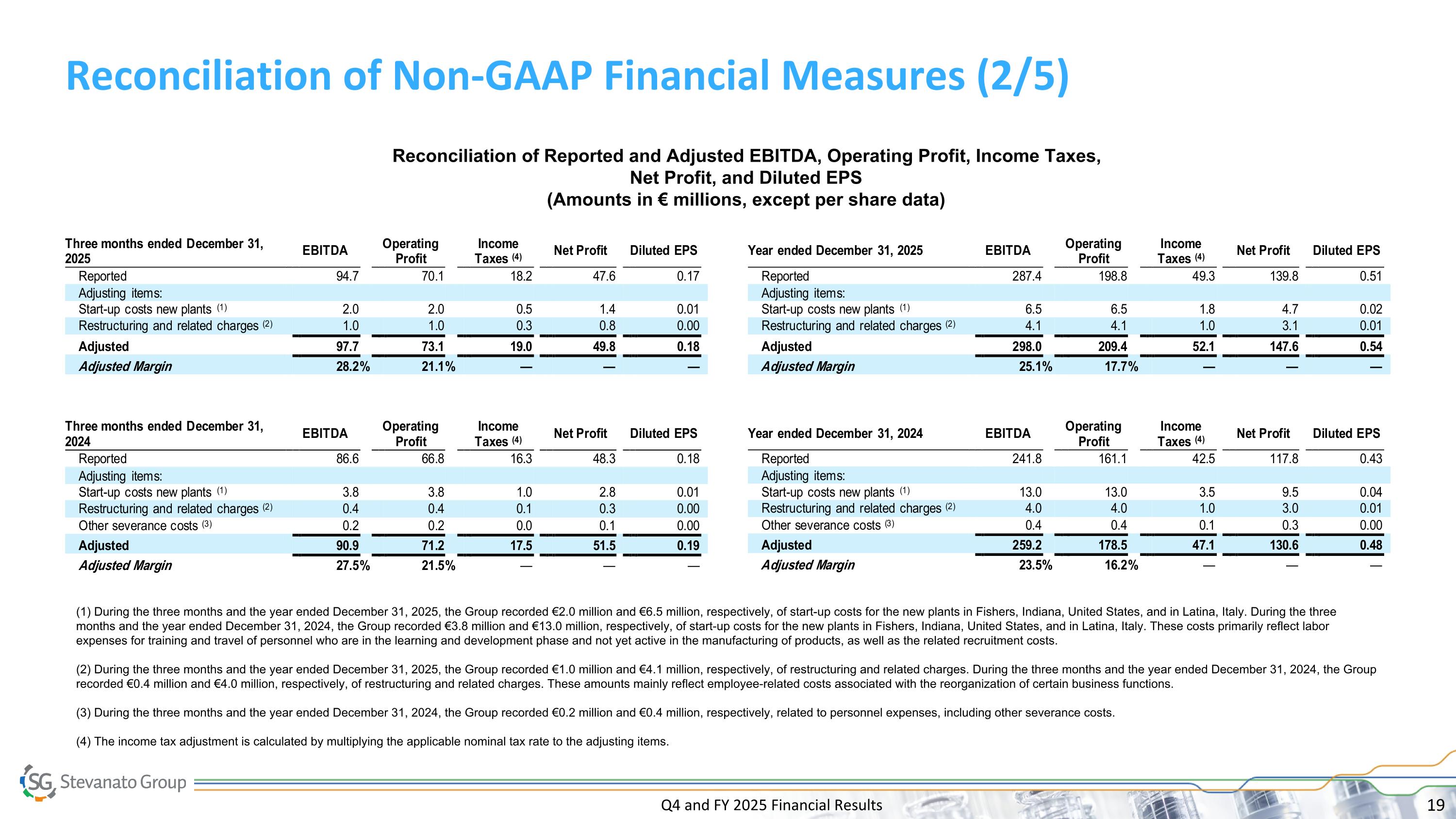
Q4 and FY 2025 Financial Results Reconciliation of Non-GAAP Financial Measures (2/5) Reconciliation of Reported and Adjusted EBITDA, Operating Profit, Income Taxes, Net Profit, and Diluted EPS (Amounts in € millions, except per share data) (1) During the three months and the year ended December 31, 2025, the Group recorded €2.0 million and €6.5 million, respectively, of start-up costs for the new plants in Fishers, Indiana, United States, and in Latina, Italy. During the three months and the year ended December 31, 2024, the Group recorded €3.8 million and €13.0 million, respectively, of start-up costs for the new plants in Fishers, Indiana, United States, and in Latina, Italy. These costs primarily reflect labor expenses for training and travel of personnel who are in the learning and development phase and not yet active in the manufacturing of products, as well as the related recruitment costs. (2) During the three months and the year ended December 31, 2025, the Group recorded €1.0 million and €4.1 million, respectively, of restructuring and related charges. During the three months and the year ended December 31, 2024, the Group recorded €0.4 million and €4.0 million, respectively, of restructuring and related charges. These amounts mainly reflect employee-related costs associated with the reorganization of certain business functions. (3) During the three months and the year ended December 31, 2024, the Group recorded €0.2 million and €0.4 million, respectively, related to personnel expenses, including other severance costs. (4) The income tax adjustment is calculated by multiplying the applicable nominal tax rate to the adjusting items.
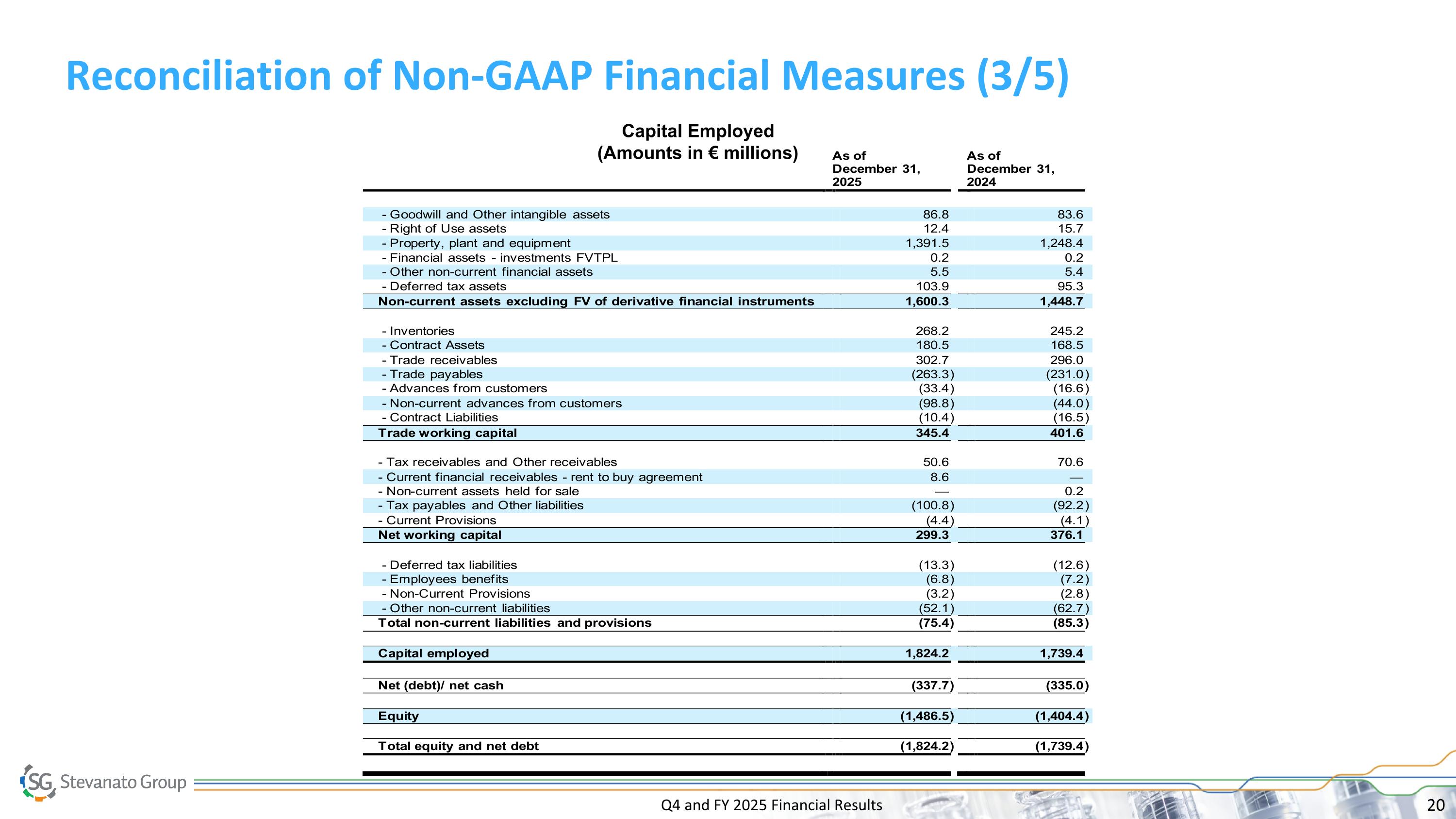
Q4 and FY 2025 Financial Results Reconciliation of Non-GAAP Financial Measures (3/5) Capital Employed (Amounts in € millions)
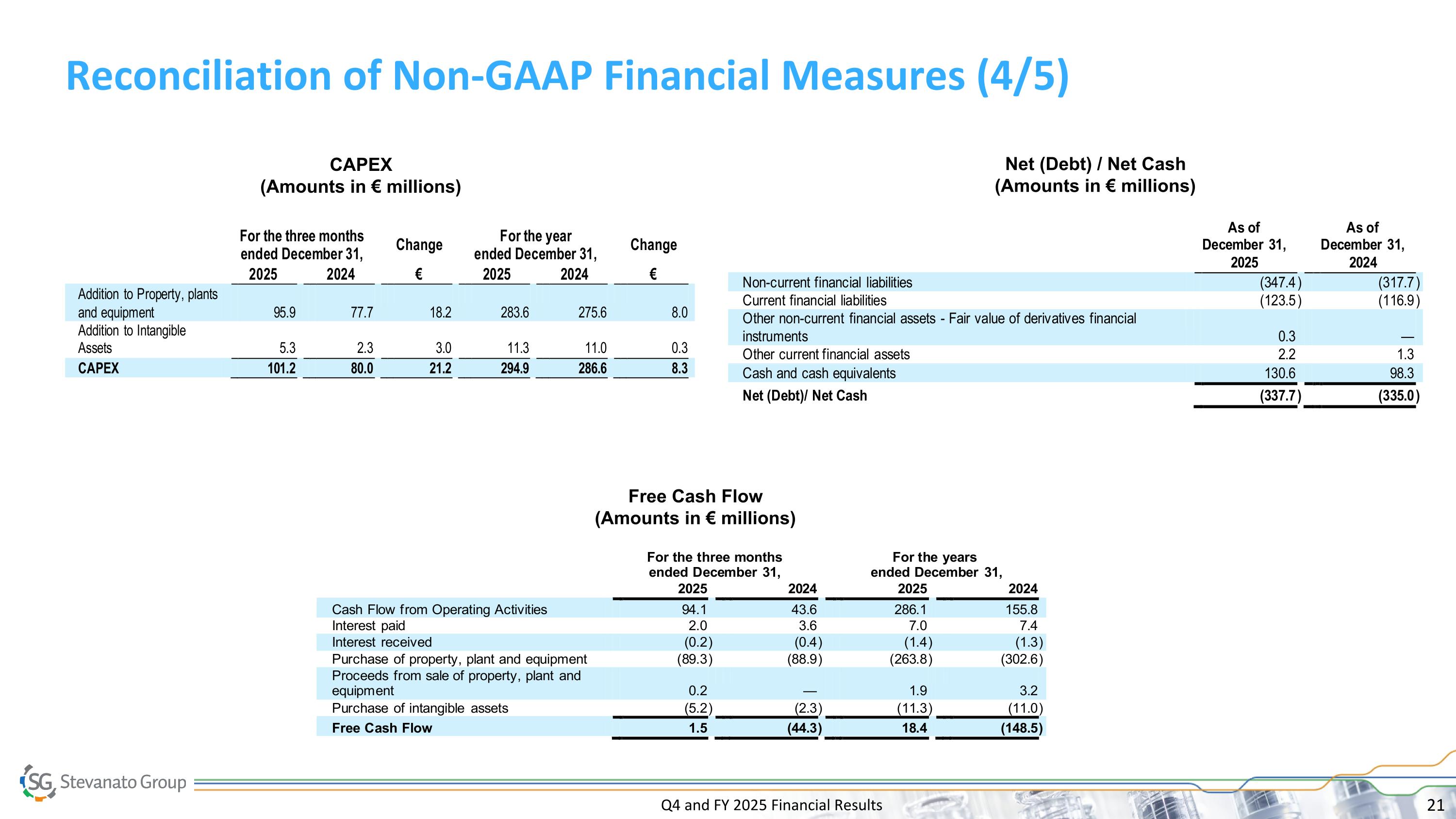
Q4 and FY 2025 Financial Results Reconciliation of Non-GAAP Financial Measures (4/5) Net (Debt) / Net Cash (Amounts in € millions) Free Cash Flow (Amounts in € millions) CAPEX (Amounts in € millions)
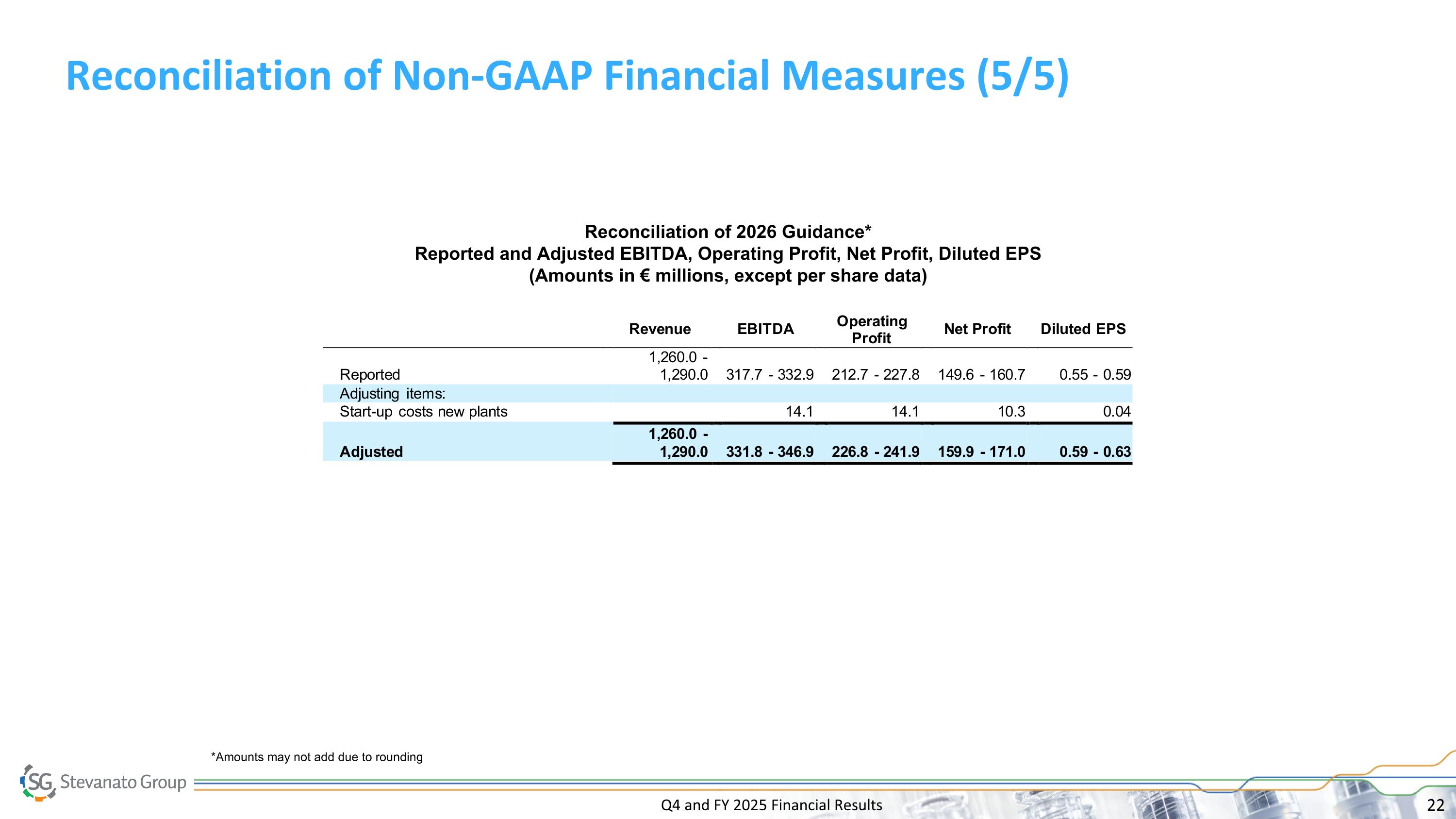
Q4 and FY 2025 Financial Results Reconciliation of Non-GAAP Financial Measures (5/5) Reconciliation of 2026 Guidance* Reported and Adjusted EBITDA, Operating Profit, Net Profit, Diluted EPS (Amounts in € millions, except per share data) *Amounts may not add due to rounding